Horn Shark

Heterodontus francisci
These small sharks are members of the bullhead family. Slow moving and nocturnal, they scavenge close to shore, and return to the same resting place each day. Both dorsal fins sport sharp spines, and should be handled with care.
Order – Heterodontiformes
Family – Heterodontidae
Genus – Heterodontus
Species – francisci
Common Names
- English: horn shark, bullhead shark, pig shark, and horned shark
- Dutch: Californische stierkophaai
- Danish: Californisk hornhaj
- Spanish: dormilón cornudo, gata, tiburón cabeza de toro, tiburón cornudo, tiburón gato, and tiburón puerco
- Czech: kalifornský žralok plochozubý
- French: requin dormeur cornu
- Finnish: sarvihai
- German: stierkopfhai
Importance to Humans
Their meat is not consumed normally and is usually discarded bycatch but can be used for fishmeal. They are seen commonly in public and private aquariums, where they can be maintained successfully for long periods (Compagno, 2001; Ebert, 2003).
Danger to Humans
Horn sharks may bite when harassed. According to the International Shark Attack File, there is one recorded instance of a shark bite involving a Heterodontid, a Port Jackson (Heterodontus portusjacksoni).
Conservation
This species is currently listed as “Data Deficient” by IUCN. But is likely “Least concern” and the populations are doing well throughout their range.
> Check the status of the horn shark at the IUCN website.
The IUCN is a global union of states, governmental agencies, and non-governmental organizations in a partnership that assesses the conservation status of species. If the gillnet fishery in Mexico expands significantly in the future, the population could potentially face problems.
Geographical Distribution
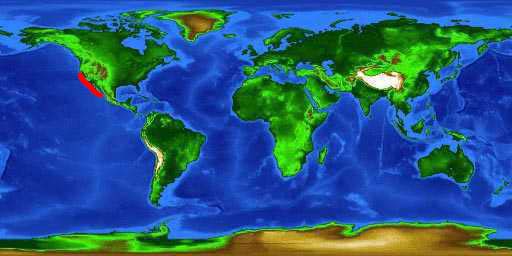
The horn shark, Heterodontus francisci, occurs in warm-temperate and subtropical regions of the eastern Pacific Ocean from central California to the Gulf of California (Compagno, 2001).
Habitat
The horn shark is common in warm-temperate to subtropical waters. Normally found along continental shelves, found as deep as 152 m (499 ft.). However, it is most commonly seen at 2-11 m (7-36 ft.), preferring 20 °C (70°F) water. During winter, they migrate to deep water, more than 30 m (98 ft.). Interestingly this species segregates by age. Adolescents between 35-48 cm (1-1.6 ft.), are normally seen in 40-150 m (131-492 ft.) on sandy bottoms. As the sharks age, they move closer and closer to shallower habitats, thus reducing competition between age groups. Adults live on rocky reefs in caves and crevices. They are nocturnal, resting in the same site from dawn till dusk every day, hunting a small area of 1,000 m² (3,300 ft²). (Roedel and Ripley, 1950; Nelson and Johnson, 1970; Feder et al., 1974; Finstad and Nelson, 1975; Strong, 1989; Ebert, 2003).
Biology

Distinctive Features
This species has a short, blunt head with high ridges above the eyes. They have spines on the 1st and 2nd dorsal fins as well as the anal fin. The first dorsal fin originates over the pectoral-fin bases. Horn sharks use their broad muscular pelvic fins as primitive feet to move along the bottom. The supraorbital ridges are moderately low and abruptly truncated posteriorly. The interorbital space is deeply concave (the depth between ridges is less than one-fourth the eye length). Their eyes also lack nictating membranes. The pre-first dorsal-fin length can be 22 to 27% of TL, while the anal-caudal space is 4 to 8%. Adult’s first dorsal-fin is moderately high and semifalcate, and are 9 to 14% of TL in height, making them slightly larger than pelvic-fins. The second dorsal fin originates over or near the pelvic-fin terminus. The second dorsal-fin is slightly falcate and roughly the same size as the first. The anal-fin is subangular and slightly falcate. (Compagno, 2001; Ebert, 2003).
Coloration
The dorsal surface dark to light grey or brown with dark brown or black spots, which are generally less than one-third eye diameter, on the body and fins. Smaller spots are particularly concentrated on head and tail. The ventral side is a yellow (Compagno, 2001; Ebert, 2003).

Dentition
Horn sharks have 19–26 tooth rows of teeth in the upper jaw and 18–29 tooth rows in the lower. Front teeth are small and pointed, sporting a central cusp with a pair of lateral cusplets. Teeth at the sides of the mouth are larger and wider, like a molar and are used for crushing (Compagno, 2001; Ebert, 2003).
Denticles
Small and smooth, adults have 200 denticles per cm² (Compagno, 2001).
Size, Age, and Growth
The maximum total length is 120 cm (4 ft) (Roedel and Ripley 1950, Feder et al. 1974). However, most adults only grow to 96 cm (3.1 ft). Females mature at roughly 58 cm (2 ft) (Compagno et al. 1995). While males are smaller and mature at 56 – 61 cm (1.8-2 ft) (Strong 1989). New born pups are 15-17 cm (6-6.7 in) (Compagno et al. 1995, Ebert 2003).

Food Habits
Adults feed almost exclusively on mollusks, echinoderms, and crustaceans. In order, the crush its well-armored prey these sharks bite down with the highest known bite force relative to size of any shark. Ranging from 95 to 200 N (Huber et al., 2005). The largest adults feed on the short-spined purple urchin, (Strongylocentrotus purpuratus) which stains their teeth and fin spines purple (Ebert, 2003). Horn sharks have far less ampullae of Lorenzini than other sharks (148), making electroreception a lesser part of their hunting strategy (Bullock et al., 2005).
Reproduction
Horn sharks are oviparous, laying eggs (usually two) every 11-14 days, between February and April. As many as 24 eggs may be laid in a single season (Ebert, 2003). Egg cases are conical and surrounded by two broad spiral flanges. This shape enables a mother to wedge the eggs into crevices, protecting them from would-be predators (Compagno, 2001). Eggs usually laid 2-13 m (7-43 ft.) deep. Development lasts 6-10 months, but can change with water temperature (Dempster and Herald 1961, Ebert 2003).
Predators
Other sharks and large fish are potential predators on horn sharks.
Taxonomy
The horn shark belongs to the family Heterodontidae (bullhead sharks). The genus was described by Blainville in 1816. The species was described by Girard in 1855. The current valid scientific name for the horn shark is Heterondontus francisci. This comes from the Greek word “heteros”, meaning different; and the Greek word “odont”, meaning teeth. One synonym used in past scientific literature referring to this species is Gyropleurodus francisci (Girard, 1854). Heterodontus californicus (Herald 1961) was used in apparent error for H. francisci.
Prepared by: Robert Buch
Revised by: Tyler Bowling 2019
References:
- Bullock, T.H., Hopkins, C.D., Popper, A.N. and Fay, R.R. 2005. Electroreception. Birkhäuser. p. 49.
- Compagno, L.J.V. 2001. Sharks of the world. An annotated and illustrated catalogue of shark species known to date. Vol. 2. Bullhead, mackeral and carpet sharks (Heterodontiformes, Lamniformes and Orectolobiformes). FAO species catalogue for fisheries purposes. No. 1. Vol. 2. FAO, Rome.
- Compagno, L.J.V., Krupp, F. and Schneider, W. 1995. Tiburones. In: W. Fischer, F. Krupp, W. Schneider, C. Sommer, K.E. Carpenter, and V.H. Niem (eds). Guía FAO para la identificación de especies para los fines de la pesca. Pacífico Centro-Oriental. Volumen II, Vertebrados-Parte 1. pp: 647–743. FAO, Roma.
- Dempster , R.P. and Herald, E.S. 1961. Notes on the hornshark, Heterodontus francisci, with observations on mating activities. Occasional Papers of the California Academy of Sciences 33: 1–7.
- Ebert, D.A. 2003. Sharks, Rays and Chimaeras of California. University of California Press, Berkley.
- Eschmeyer, W.N., Herald, E.S. and Hammann, H. 1983. A field guide to Pacific coast fishes of North America. Houghton Mifflin Company, Boston, USA.
- Feder, H.M., Turner, C.H. and Limbaugh, C. 1974. Observations on fishes associated with kelp beds in southern California. California Fish and Game Fish Bulletin 160: 44.
- Finstad, W.O. and Nelson, D.R. 1975. Circadian activity rhythm in the horn shark, Heterodontus francisci: effect of light intensity. Bulletin of the Southern California Academy of Science. 74: 20–26
- Huber, D.R., Eason, T.G., Hueter, R.E. and Motta, P.J., 2005. Analysis of the bite force and mechanical design of the feeding mechanism of the durophagous horn shark Heterodontus francisci. Journal of Experimental Biology, 208(18), pp.3553-3571.
- Michael, S.W. 1993. Reef sharks and rays of the world. A guide to their identification, behavior and ecology. Sea Challengers, Monterey, California.
- Nelson, D.R. and Johnson, R.H. 1970. Diel activity rhythms in the nocturnal, bottom-dwelling sharks, Heterodontus francisci and Cephaloscyllium ventriosum. Copeia 1970(4): 732–739.
- Strong, W.R. 1989. Behavioral ecology of horn sharks, Heterodontus francisci, at Santa Catalina Island, California, with emphasis on patterns of space utilization. California State University, Long Beach. M.S. Thesis
- Roedel, P.M. and Ripley, W.E. 1950. California sharks and rays. California Fisheries Bulletin No. 75.