The iconic eyespots that some moths and butterflies use to ward off predators likely evolved in distinct ways, providing insights into how these insects became so diverse.
A new study manipulated early eyespot development in moth pupae to test whether this wing pattern develops similarly in butterflies and moths. The results suggest that the underlying development of eyespots differs even among moth species in same family, hinting that moths and butterflies evolved these patterns independently.
Influencing how eyespots form can lead to a better understanding of the respective roles genetics and the environment play in moth and butterfly wing patterns, said lead author Andrei Sourakov.

“Moths stumbled on a very successful evolutionary design over 200 million years ago,” said Sourakov, collections coordinator of the Florida Museum’s McGuire Center for Lepidoptera and Biodiversity. “That’s a long time for evolution to take place. It’s easy to assume that things that look the same are the same. But nature constantly finds a way of answering the same question with a different approach.”
Sourakov and co-author Leila Shirai, a biologist at the University of Campinas in Brazil, analyzed eyespot development in io and polyphemus moths, two species in the Saturniidae family. The eyespots in the two species responded differently to the study’s treatments, though the findings suggest the same signaling pathways were active. The researchers also found moths’ wing pattern development, which begins when they are caterpillars, slows just after they enter their pupal stage, a finding that echoes previous butterfly research.
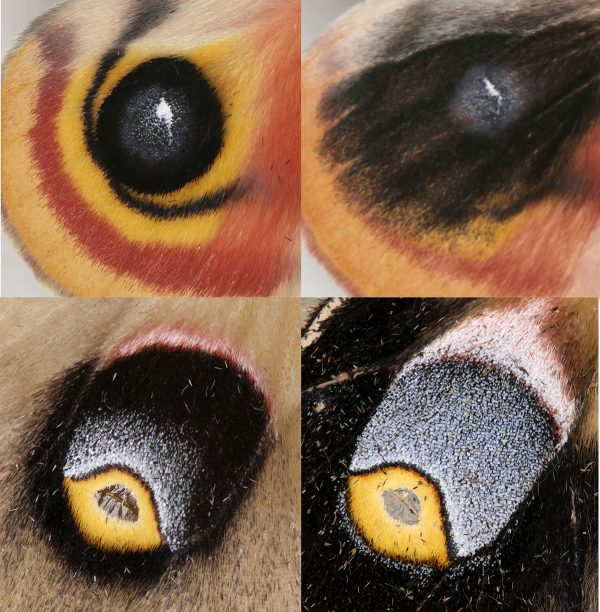
Honing in on the signaling pathways involved in eyespot development – the molecular cascade that produces pigmentation and pattern in moths and butterflies – is central to determining the similarities and differences between moth and butterfly development, Sourakov said. Looking at DNA isn’t enough. Instead, scientists need to determine what happens after a gene is expressed to see if seemingly identical wing patterns truly are the same.
“Genetically controlled variation can look identical to environmentally induced variation,” Sourakov said. “Variation isn’t really produced by genes themselves, but by the intermediate product of the gene – in this case, molecular pathways.”
Sourakov and Shirai’s research expands on a 2017 study by Sourakov that showed molecules in the blood thinner heparin influenced eyespot development in moths.
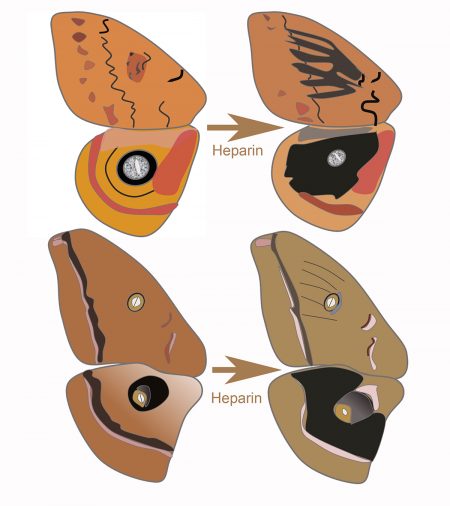
In the new study, heparin triggered various changes in moth eyespots, including smudging and a shift in proportion. Despite similar molecular interactions, however, the changes were inconsistent between the io and polyphemus moths, potentially due to the different ways their wing patterns are mapped out by genes.
Sourakov and Shirai were able to detect wing development was likely paused just after pupation by delivering varying doses of heparin to caterpillars and pupae at different developmental stages. They also found eyespot tissue transplanted to a different region of the wing during pupation could induce patterning.
Natural history collections are key resources in revealing which wing patterns took hold genetically and became visible in populations, Sourakov said.
“Collections are where it all starts and where it all ends, frankly,” he said. “We can generally look at collections as a window into evolution, helping us understand which changes are just lab results and which ones can actually be observed in nature. Variation in genetics and physical characteristics is the toolbox for the evolution of diversity, and diversity is what we study at the museum. Collections help us understand that.”
The study was published in Tropical Lepidoptera Research.
Funding for the research was provided by the Sao Paulo Research Foundation and the Center for Systematic Entomology.
Source: Andrei Sourakov, asourakov@flmnh.ufl.edu